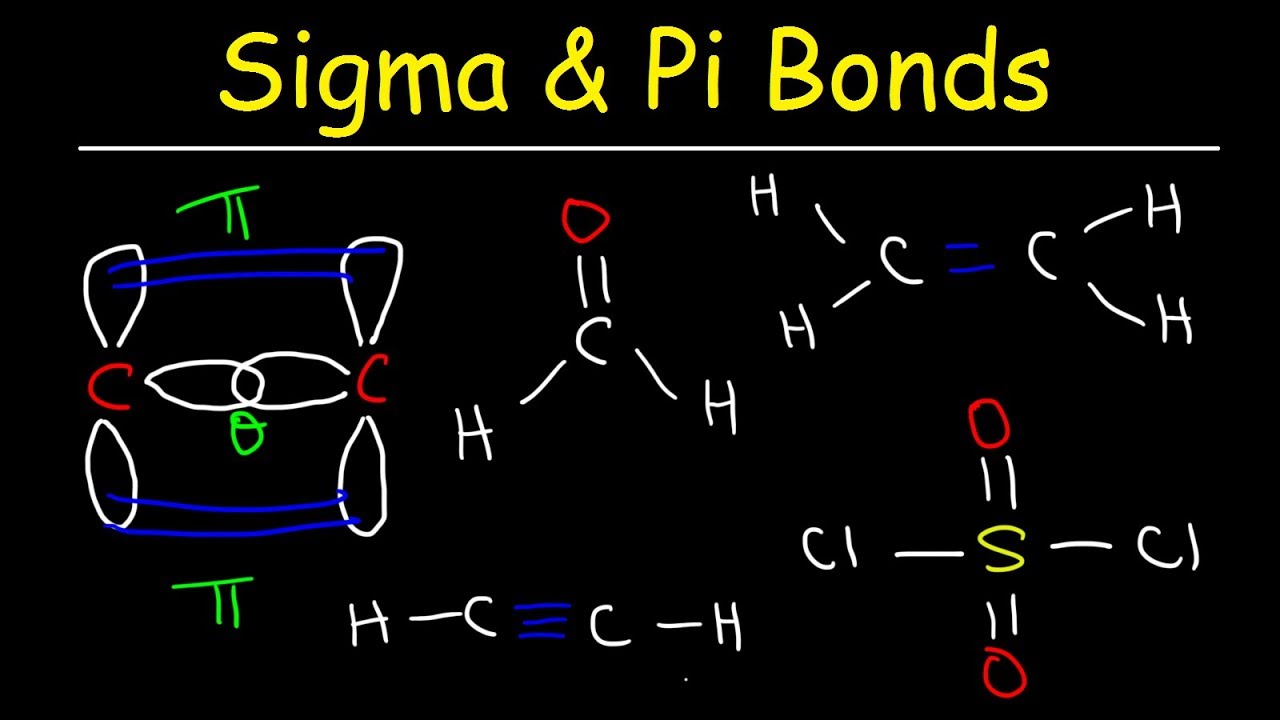
This chemistry video tutorial provides a basic introduction into sigma and pi bonds. It explains how to calculate the number of sigma and pi bonds in a molecule given its lewis structure. It contains plenty of examples and practice problems.
My E-Book:
Video Playlists:
Homework Help:
Subscribe:
Support & Donations:
Youtube Membership:
New Chemistry Video Playlist:
Disclaimer: Some of the links associated with this video may generate affiliate commissions on my behalf. As an amazon associate, I earn from qualifying purchases that you may make through such affiliate links….(read more)
LEARN MORE ABOUT: Treasury Inflation Protected Securities
REVEALED: Best Investment During Inflation
HOW TO INVEST IN GOLD: Gold IRA Investing
HOW TO INVEST IN SILVER: Silver IRA Investing

This is beautiful
You are amazing❤
Thanks a lot. I just learned it it so easily❤❤
Noooo my kryptonite….
Honestly watching these videos makes me wonder what the point of paying to go to multiple hour long college lectures every week when i can just watch some 6 minute youtube video that explains it perfectly for me
Absolutely astonishing I learnt this in less than 30 seconds. You are truly amazing.
My guy skipped L in the alaphabet
The first 30 seconds of this video helped me more than my hours of college lecture. Give this man a medal.
You a Master bro
This video summarized pretty much everything I need to know and the practice questions were really helpful, you made me a lot more confident using this video, words cannot express how grateful I feel right now ❤
How can I say thank you my teacher❤️
u are literally 5000 times better than my chem professor, thank u so much sir!!
Please,can you transate these videos for arabic
Thank you sir
you say sigma but you write theta
Thank you for your videos…
what's the hybridization he talks abt at the beginning
Credit goes to YouTube Recommendations ❣️
1:55 lmao we just got taught the other day that in double bond 1 is pi and other is sigma how is this possible that there is 2 pi bond
you have such a nice voice, can we get married
Best youtube professor <3
Thank you so much. I was just struggling with the topic and was having a mental breakdown but then I found your video and I just understood everything in 6 min after one hour struggle… so thank you soooooooooo much .
1:50 but if the unhybridized p orbitals are empty then how do they have forces attracting them? is it just that its random chance that an electron might leave one of its sigma bonds and travel up there? wouldnt that weaken or destroy the sigma bond since theres limited electrons?
I'm absolutely amazed at how fast I learned that. Thank you!
I literally learned exactly what I needed to in first 20 seconds….
ive never felt a video to be as personalized to me as this one
got exactly what i needed, nothing more, nothing less in like 6 min
@5:33, what is the IUPAC name of the structure drawn?
Thank you soo much this is soo helpful god bless you ❤❤❤
You are best chemistry teacher on YouTube
You're a life saver….your videos always help me get concepts
WHA this is so easy ???
Good lecture may I have notes in written form?
My teacher is literally so confusing and she thanks to you I know this stuff
bro your voice is so calming you gon make me act up
Thank you for your service
2 minutes are enough to know the whole concept
Thank you very much mister. This is really help me
Never knew it was this ez thanks man